Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
By A Mystery Man Writer
Description

Determine Compressibility of Gases

PV Compressibility factor Z= nRT is plotted against pressure : N. Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
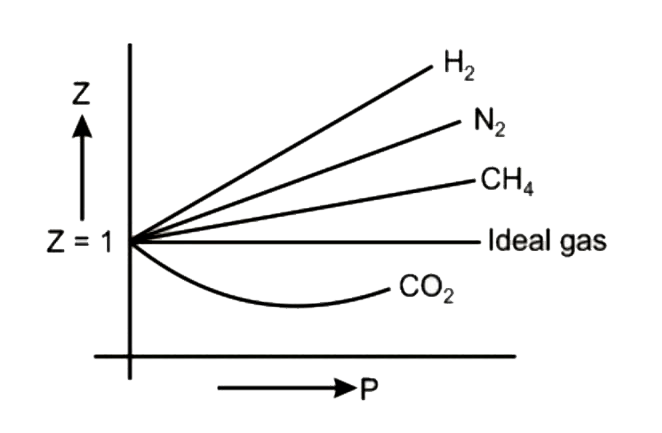
Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
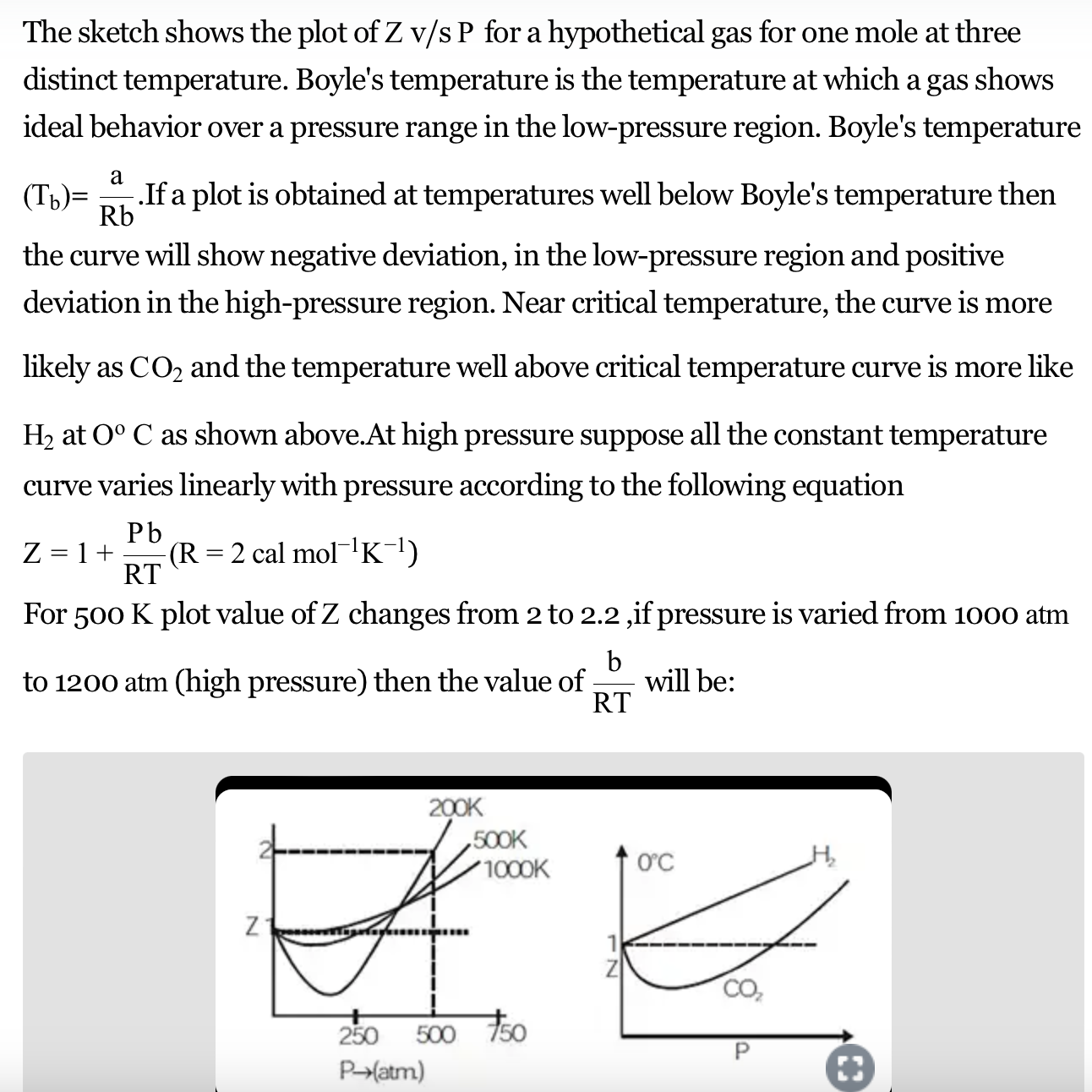
Solved The graph of compressibility factor (Z)v/sP for 1 mol

Compressibility factor - Wikipedia

UNE Uption is correct. Compressibility factor Z=- is plotted against pressure: nRT Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H

Compressibility factor - Wikipedia

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

A real gas M behaves almost like an ideal gas. Graph 1 is obtained by plotting volume, V against temperature, T for x mol of gas M at pressure, P_1. a. Suggest

Compressibility factor (Z = (P V/n R T)) is plotted against pressure  What is the correct order of liquefiability of the gases shown in the
What is the correct order of liquefiability of the gases shown in the

संपीड्यता कारक (Z=(pV)/(nRT)) को दाब के विपरीत आलेखित किया जाता है।

gas laws - Does the amount of a gas increase with pressure? - Chemistry Stack Exchange

Telugu] Compressibility factor (Z = (PV)/(nRT)) is plotted against p
from
per adult (price varies by group size)






