10.9: Real Gases - Deviations from Ideal Behavior - Chemistry LibreTexts
By A Mystery Man Writer
Description
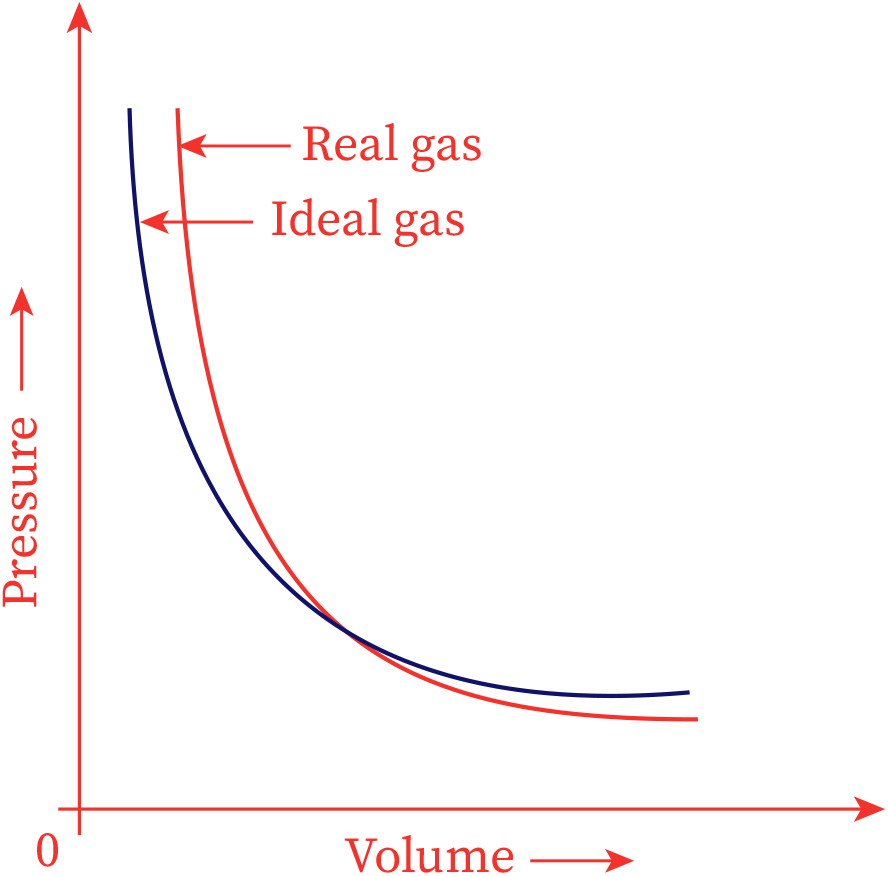
No real gas exhibits ideal gas behavior, although many real gases approximate it over a range of conditions. Gases most closely approximate ideal gas behavior at high temperatures and low pressures. …
No real gas exhibits ideal gas behavior, although many real gases approximate it over a range of conditions. Gases most closely approximate ideal gas behavior at high temperatures and low pressures. Deviations from ideal gas law behavior can be described by the van der Waals equation, which includes empirical constants to correct for the actual volume of the gaseous molecules and quantify the reduction in pressure due to intermolecular attractive forces.
No real gas exhibits ideal gas behavior, although many real gases approximate it over a range of conditions. Gases most closely approximate ideal gas behavior at high temperatures and low pressures. Deviations from ideal gas law behavior can be described by the van der Waals equation, which includes empirical constants to correct for the actual volume of the gaseous molecules and quantify the reduction in pressure due to intermolecular attractive forces.

1.3 Deviation from ideal gas behaviour

AP Chemistry 2024 – 3.6 Deviation from the Ideal Gas Law
Ideal and Real Gases
1. An unspecified ideal gas at 10°C and 100kPa occupies a volume of 2.5 m³. (a) How many moles does this gas have? (b) If the pressure and temperature are raised three

Real gases: Deviations from ideal behavior, AP Chemistry

Chemistry 103-Principles of Chemistry PDF, PDF, Chemical Compounds
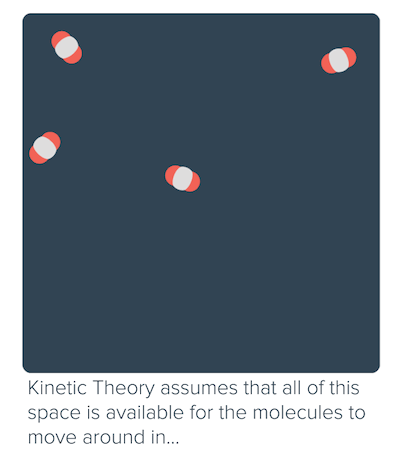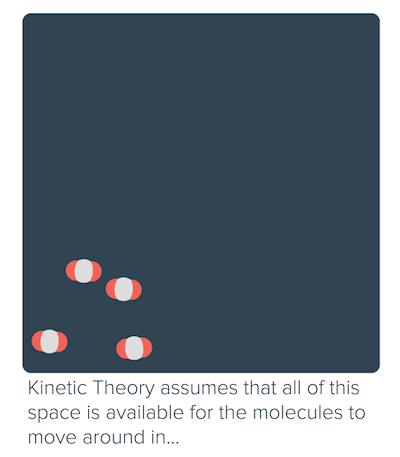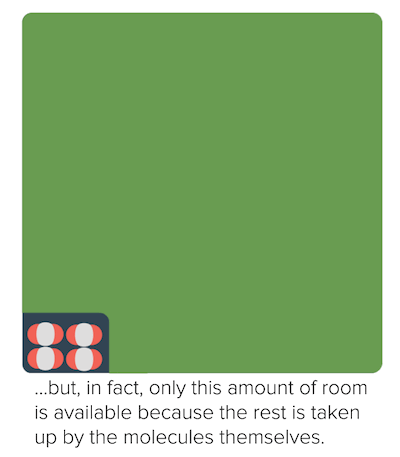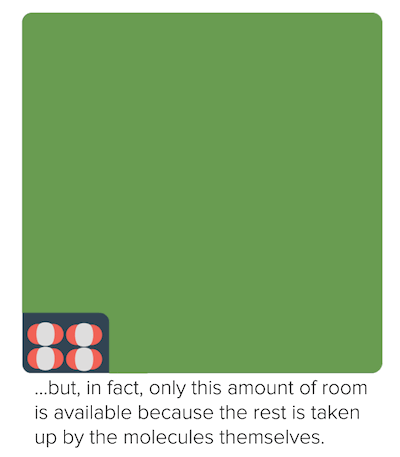
Non-ideal behavior of gases (article)

Solved Which statement about real vs. ideal gases is not

Chapter 10 - Gases Prof. Dr. Nizam M. El-Ashgar - ppt download

10.8: Molecular Effusion and Diffusion - Chemistry LibreTexts

Real gases: Deviations from ideal behavior, AP Chemistry
10.8: Molecular Effusion and Diffusion - Chemistry LibreTexts
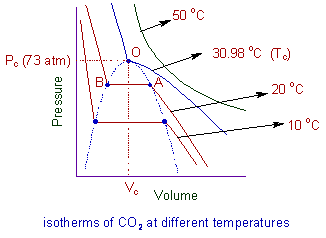
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR
from
per adult (price varies by group size)
:max_bytes(150000):strip_icc()/Health-Ways-to-Lower-Cortisol_Horiz-3b2f5e9ccace4c20b1416d36c0f93914.jpg)






