What is the mass of glucose required to produce 44g of C{O_{2'}} on complete combustion?30g45g60g22g
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:what is the mass of glucose required to produce 44g of co2 on complete
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g

SOLVED: What mass of ethanol can be produced from 2.27 x 10^9 g of glucose if the yield is 85.0%? C6H12O6 â†' 2 C2H5OH + 2 CO2 (g) Molar Mass: C6H12O6 =
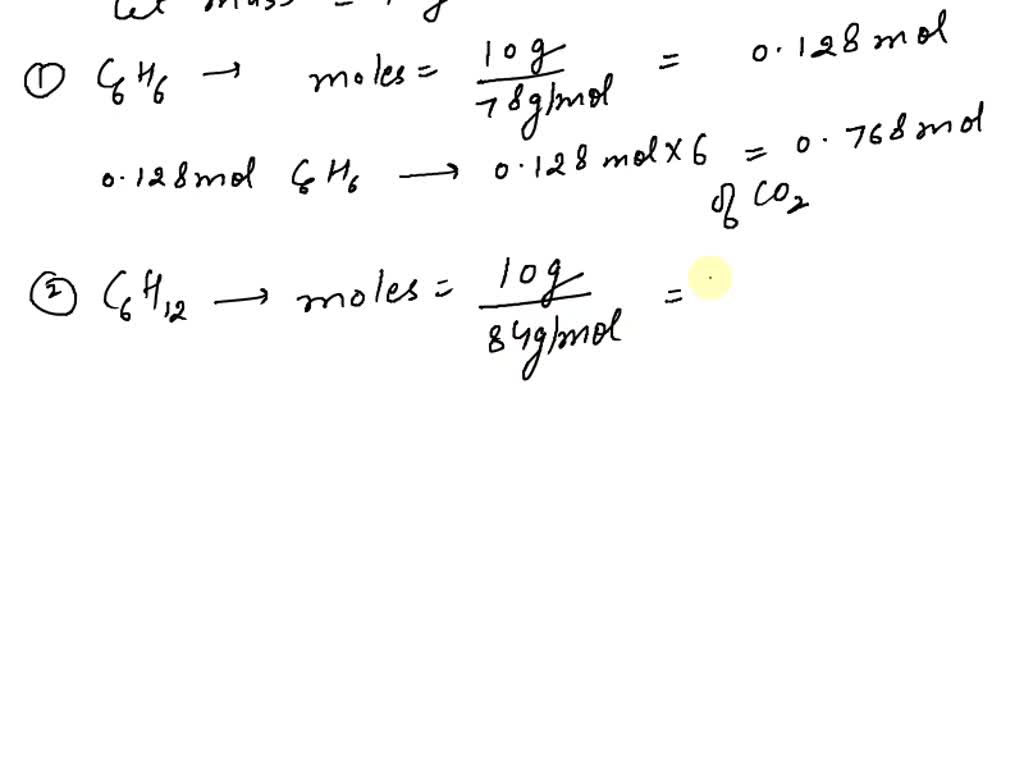
SOLVED: If equal masses of the following compounds undergo complete combustion, which will yield the greatest mass of CO2? 1. Benzene, C6H6 2. Cyclohexane, C6H12 3. Glucose, C6H12O6 4. Methane, CH4

What is the mass of glucose required to produce 44g of Co2 on complete combustion

SOLVED: If equal masses of the following compounds undergo complete combustion, which will yield the greatest mass of CO2? 1. Benzene, C6H6 2. Cyclohexane, C6H12 3. Glucose, C6H12O6 4. Methane, CH4

PPT - Stoichiometry 1 Formulas and the Mole PowerPoint Presentation, free download - ID:2368206

Solved Consider the combustion of 5.000 g of glucose
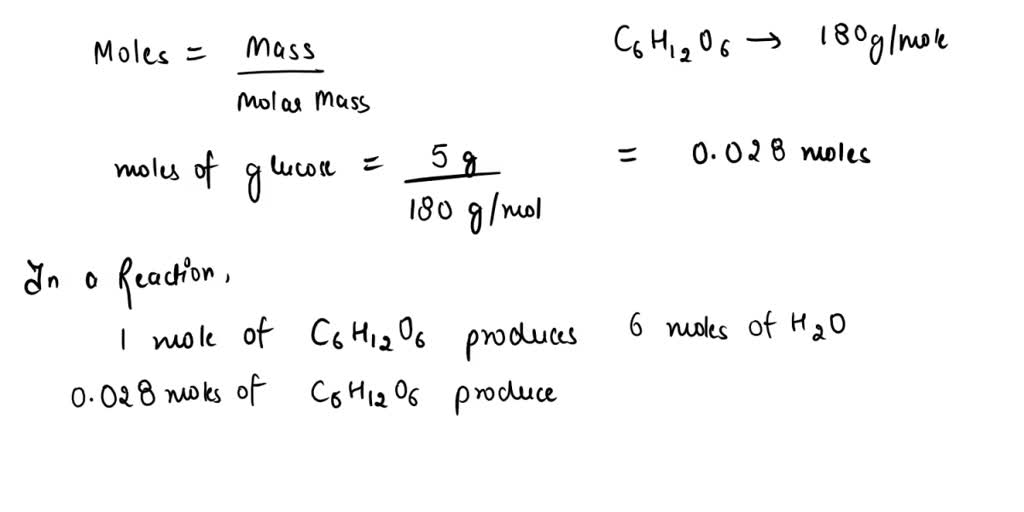
SOLVED: How many grams of water are produced in the combustion of 5.00g of glucose, C6H12O6? C6H12O6(s) + 6 O2(g) â†' 6 CO2(g) + 6 H2O(l)

Solved -. The atomic mass of oxygen (O2) is 16.00 g/mol.

Solved Glucose, also known as blood sugar, has the

Determine the mass of CO2 produced by burning enough of each fuel

what is the mass of glucose required to produce 44grams of CO2 on complete combustion - 10ijyqww

Solved The food we eat is degraded, or broken down, in our
from
per adult (price varies by group size)







-10755-p.jpg)