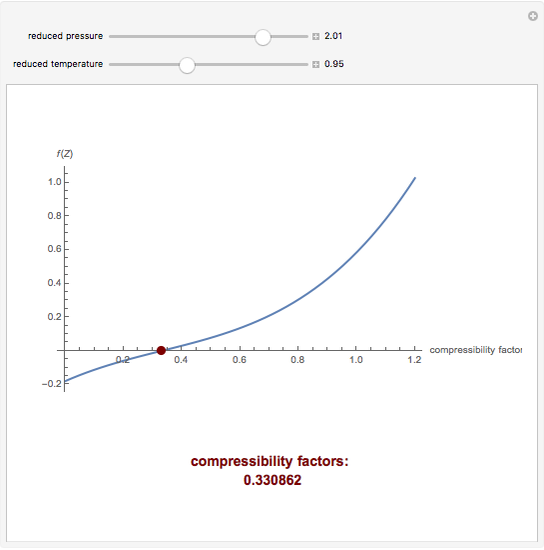
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
By A Mystery Man Writer
Description
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
What is the value of compressibility factor in terms of vander waal cons-an t at different conditions of pressure and volume-Why is Z-1 for H2 and He gas
What is the value of compressibility factor in terms of vander waal cons-an t at different conditions of pressure and volume-Why is Z-1 for H2 and He gas

If Z is a compressibility factor, van der Waals equation at low pressure ..
Slope of graph of compressibility factor(Z) with pressure(P) for hydrogen gas at any pressure i

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

Compressibility factor - Wikipedia
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
Why does the van der Waals equation have one positive and one negative correction term? - Quora

Gas compressibility factor Z: Ideal gas vs Real gas
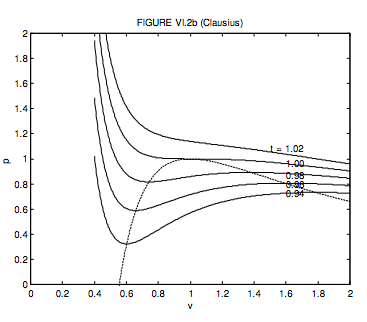
6.3: Van der Waals and Other Gases - Physics LibreTexts

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application
from
per adult (price varies by group size)