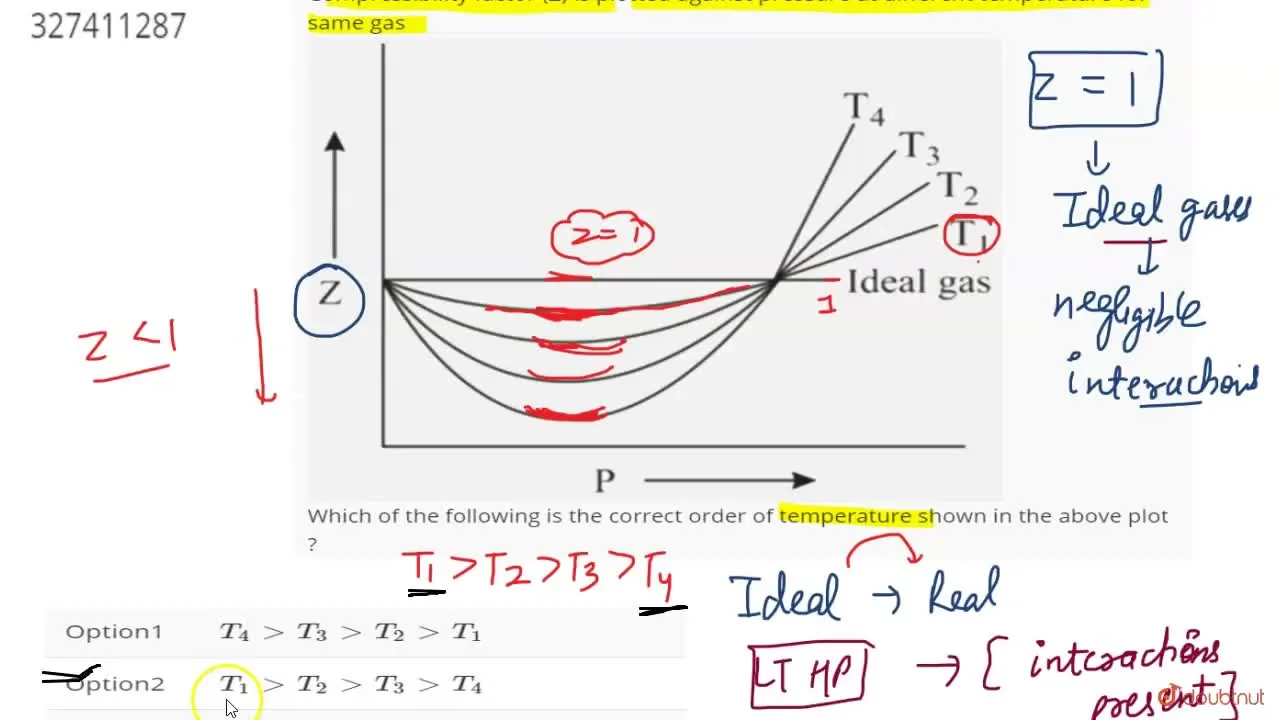
For a given gas, a graph is shown between compressibility factor (Z) and Pressure (P).Select the incorrect statement(s) about the various temperature relations.a)Temperature T1 must be above critical temperature (TC).b)Temperature T2 may
By A Mystery Man Writer
Description

Compressibility Factor - an overview

A graph Z vs P is plotted N_2 gas different temperatureThe correct relationship between temperatures

Compressibility factor Z is plotted against pressure P for four different gases A , B , C & D. The correct order of critical temperature of the gases shown in the below

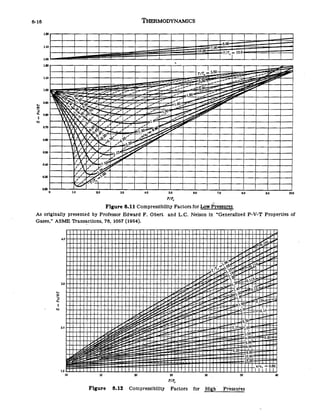
Compressibility factor (Z) is plotted against pressure at different te
Compressibility factor (Z) is plotted against pressure at different te

The given graph represents the variation of Z(compressibility factor =displaystyle frac{mathrm{P}mathrm{V}}{mathrm{n}mathrm{R}mathrm{T}}) versus mathrm{P}, three real gases mathrm{A}, mathrm{B} and C. Identify the only incorrect statement.For the gas C

compressibility Factor v/s Pressure/ Temperature Graph . States of Matter Class XI.

Compressibility factor Z is plotted against pressure p for four different gases A,B, C D. The correct order of critical temperature of the gases shown in the above graph is

The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?
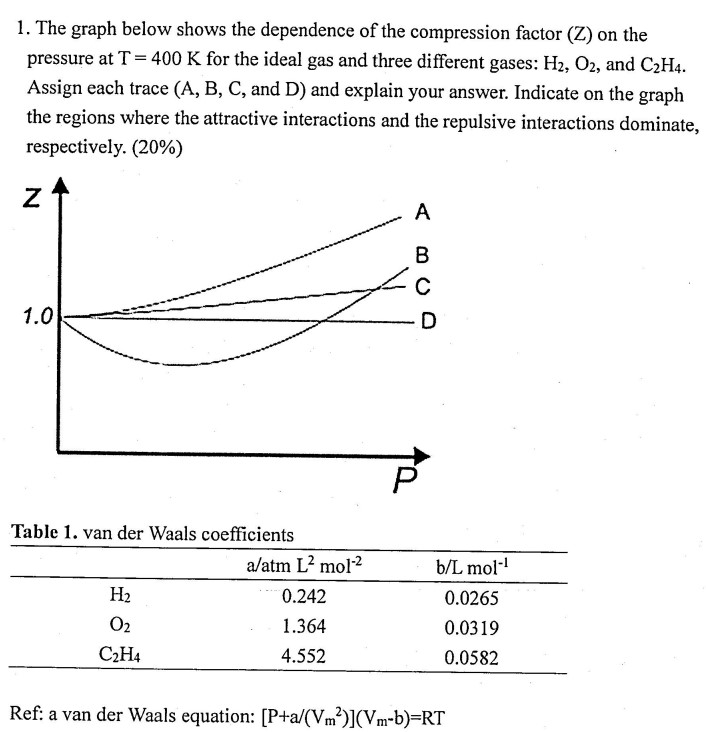
Solved The graph below shows the dependence of the

Solved The graph of compressibility factor (Z)v/sP for 1 mol

1.7: Connecting the van der Waals and the viral equations- the Boyle temperature - Chemistry LibreTexts
from
per adult (price varies by group size)