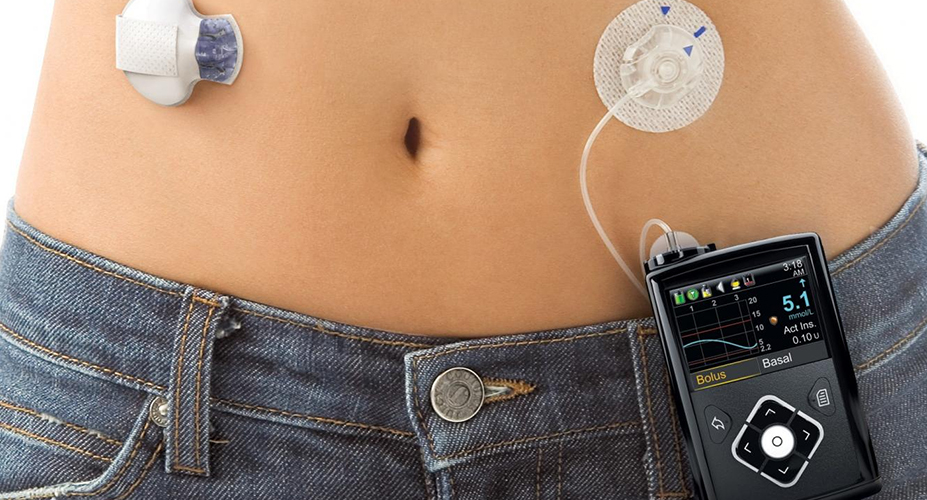
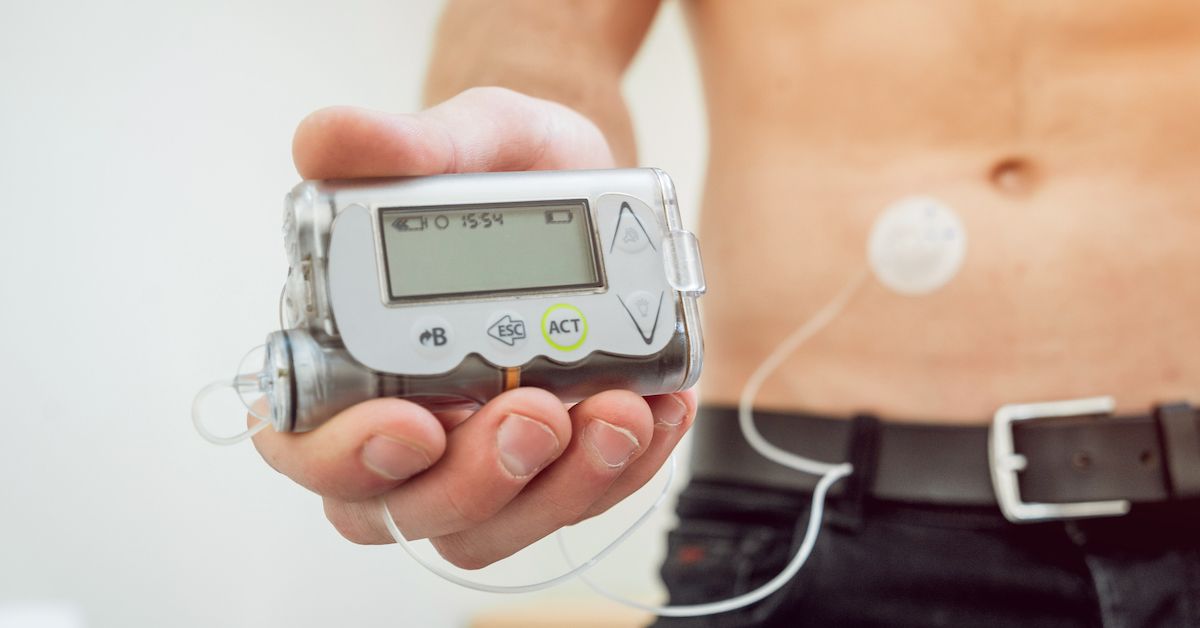
Medtronic recalls some insulin pumps that could lead to dangerous incorrect dosing
By A Mystery Man Writer
Description
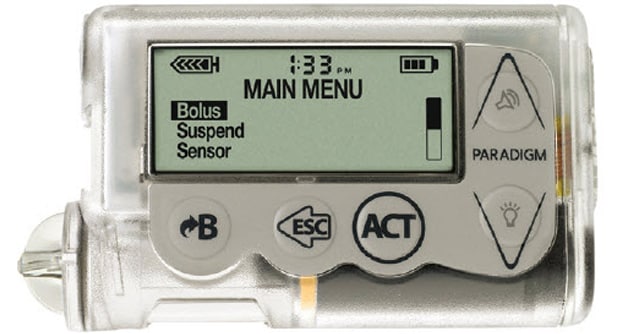
Medtronic is recalling MiniMed 600 Series Insulin Pumps. The FDA calls recall Class I the most serious type of recall, which can lead to injury or death.
This is additional taxonomy that helps us with analytics
This is additional taxonomy that helps us with analytics

FDA says Medtronic MiniMed insulin pump recall is serious - MassDevice

Medtronic Announces FDA Approval for MiniMed 770G System

Insulin pumps have most reported problems in FDA database

Different Reviews: Medtronic 670G

I Lost My Spouse Because an Insulin Pump Was Defective

Deep flaws in FDA oversight of medical devices, and patient harm, exposed in lawsuits and records
Medtronic Recalls Mini Med and Pro Infusion Insulin Pumps due to Life-Threatening Injuries
media./photos/5d28fe551508cc000882244e/4

FDA Product Recall: Medtronic Recalls MiniMed Insulin Pumps for Incorrect Insulin Dosing
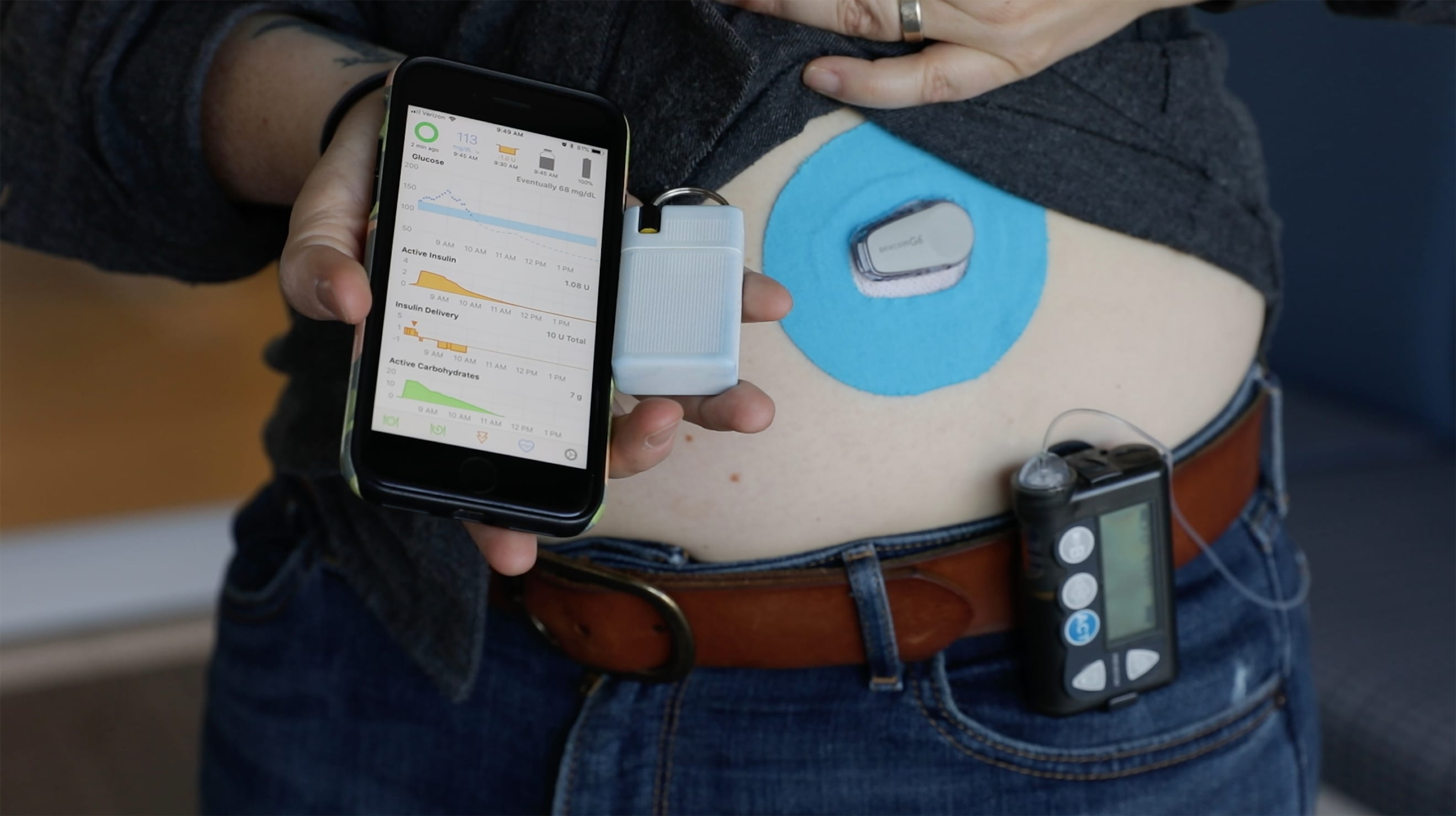
Explaining active insulin

Insulin pumps linked to more reports of injury and death than any other medical device, records show
Medtronic recalls some insulin pumps as FDA warns they can be hacked
Insulin pumps, widely used, also have the most problems in FDA's database
Medtronic recalls some insulin pumps that could lead to dangerous incorrect dosing

FDA recalls MiniMed insulin pumps
from
per adult (price varies by group size)